Journal of
eISSN: 2373-6410


Case Report Volume 3 Issue 3
1Department of Neurology, Ibn Sina Hospital, Kuwait
2Department of Medicine, Kuwait University, Kuwait
Correspondence: Ismail Ibrahim Ismail, Department of Neurology, Ibn Sina Hospital, Safat, Kuwait
Received: October 21, 2015 | Published: November 19, 2015
Citation: Al-Hashel JY, Ismail II, John JK, et al. Neurological, hematological and dermatological manifestations of vitamin b12 deficiency secondary to pernicious anemia; a case report and literature review. J Neurol Stroke. 2015;3(3):1-6. DOI: 10.15406/jnsk.2015.03.00095
Background: Vitamin B12 is a water-soluble essential vitamin. It is essential for the rapid synthesis of deoxyribonucleic acid (DNA) during cell division and thus formation of red blood cells and maintenance of a healthy nervous system as well as the skin. It is found mainly in food of animal origin (e.g. meat, eggs, and dairy products and lacks a reliable plant source. Deficiency occurs due to either poor intake or decreased absorption. Among the most common causes are: pernicious anemia, atrophic gastritis, celiac disease or following gastric surgery. Deficiency of this essential vitamin can lead to potentially irreversible neurological, psychiatric, hematological and/or dermatological symptoms.
Case Summary: We report a case of a 46-year-old omnivorous male patient who presented with gradual onset of weakness and spasticity of both lower limbs and upper limbs, parasthesia of all limbs with loss of joint position and vibration sensation. These symptoms were associated with memory changes, weight loss and hyperpigmentation of the skin. Magnetic resonance imaging (MRI) showed a long demyelinating lesion in the cervical spinal cord. Extensive laboratory investigations were performed that confirmed vitamin B12 deficiency and ruled out other common mimics associated with that condition. He had marked improvement of all his neurological and non-neurological symptoms following vitamin B12 supplementation with complete resolution of the cervical demyelinating lesion within 3 months.
Conclusion: Vitamin B12 deficiency is a common but under-diagnosed clinical condition that needs to be highlighted. It can present with neurological, psychiatric, hematological and/or dermatological manifestations. It is uncommon to see the whole systems affected together early in the course of the disease. Early diagnosis and treatment may reverse potentially permanent symptoms and signs and can prevent the disability that would otherwise ensue if left untreated. Pernicious anemia is one of the most common causes of vitamin B12 deficiency and should be ruled out in all cases even if autoantibodies against intrinsic factor and parietal cells are negative.
Keywords:Vitamin B12, Cobolamin, Methylmalonic Acid, Homocysteine, Megaloblastic Anemia, SCD (subacute combined degeneration), Pernicious Anemia
Patients with vitamin B12 deficiency can present to different specialties with different types of symptoms. Unless kept in mind in the differential diagnosis, this condition can be easily missed. Deficiency of this vitamin can negatively affect the brain and blood cells as well as the hair and skin. One of the most common causes is Pernicious Anemia (PA) which is a disease that can be fatal if untreated. It has been an area of interest long time ago to the extent that a Nobel Prize was awarded in the past century for the discovery of effective treatment.1
A 46-year-old, Kuwaiti, overweight, omnivorous male, with no family history of autoimmune disease, presented with a subacute progressive course of lower limb numbness, gait abnormality, sense of imbalance, weakness and stiffness of both lower limbs of one month duration prior to admission. Weakness and numbness progressed to involve both upper limbs within 2 weeks of the onset.
He had a history of chronic gastritis for 5 years with recurrent H. pylori infection for which he had received recurrent courses of antibiotics in the form of amoxicillin, and clarithromycin with omeprazole. Upper GI endoscopy was performed previously and showed atrophic gastritis and multiple gastric ulcers.
He, also had generalized fatigue, muscle aches, poor food intake with significant weight loss (20kg in 2 years) and heart palpitations. His family also noted a recent decline in his memory with poor attention and concentration. They also noted the darkening of his skin in the past 6 months.
There were no associated bulbar or sphincteric symptoms. There was no history of any psychiatric symptoms and he denied any history of alcohol intake. There was no history of fever, arthralgia or orogenital ulcers and no history of previous gastric surgeries.
General examination showed average body build with Body Mass Index (BMI) of 22. Hyperpigmentation of the skin was evident, especially on the forehead and face with blotchy areas of darkening all over the body (Figure 1,2).
His hair was brittle and thinned. His tongue was normal and there was no jaundice. Neurological examination showed a conscious and oriented patient yet sleepy with poor attention and concentration. His Mini Mental State Examination (MMSE) was 26 with normal fluent speech.
The cranial nerve examination was normal with bilateral normal fundi and visual acuity. Motor examination showed weakness and spasticity of both upper and lower limbs with more involvement of the lower limbs. Power was grade (4/5) in both upper limbs, grade (4-/5) in both lower limbs according to Medical Research Council (MRC) grading. All deep tendon reflexes (DTRs) in the upper limb and bilateral knee reflexes were 2+, ankle reflex was 1+ bilaterally. Plantar response was extensor on both sides. Sensory examination showed normal superficial sensations and loss of joint position and vibration sense in the lower limbs. Co-ordination was intact. Gait was ataxic-spastic.
MRI of the cervical spine showed long hyperintense cervical lesion extending from the Cervicomedullary junction to C7, mainly in the dorsal column (Figure 3,4). MRI Dorsal, Lumbosacrsal spine and Brain were normal.
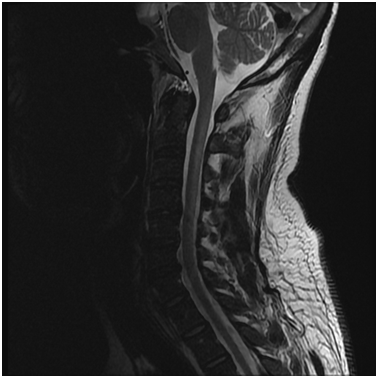
Figure 3 MRI Cervical spine, T2-weighted image (sagittal view) showing a long hyperintense lesion extending from cervicomedullary junction till C7 affecting the dorsal aspect (white arrows).
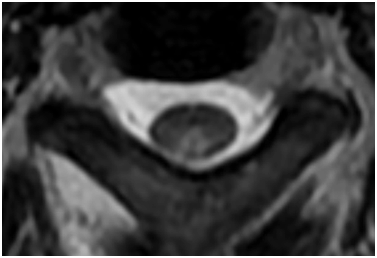
Figure 4 MRI Cervical spine, T2-weighted image (axial view) showing hyperintense lesion affecting the dorsal column (white arrow).
Nerve conduction studies (NCS) and Electromyography (EMG) showed no evidence of peripheral neuropathy.
Visual and Somatosensory evoked potentials (VEP and SSEP) were abnormal while Brainstem auditory response (BAER) was normal.
The initial laboratory assessment showed macrocytic anemia, mild leucopenia with normal renal function, liver function, electrolytes, thyroid function tests and bilirubin. Blood film showed macro-ovalocytosis, anisocytosis and hypersegmentation of neutrophils. Reticulocytic count was normal. Low Vitamin B12 level with high Homocysteine, methyl malonic acid (MMA) and lactate dehydrogenase (LDH) (Table 1).
|
Before treatment |
After treatment |
Hb |
10.3 g/dL |
13 g/dL |
MCV |
135 fL |
90 fL |
WBCs |
3.150 cells/L |
6.5 cells/L |
Vitamin B12 |
150 pg/ml |
250 pg/ml |
Homocysteine |
84.8 μmol/L |
12 μmol/L |
Methylmalonic acid |
9.6 μg/dl |
3.5 μg/dl |
LDH |
749 U/L |
150 U/L |
Table 1 Hb, Hemoglobin; LDH, Lactate Dehydrogenase; MCV, Mean Corpuscular Volume; WBCs, White Blood Cells
Serum Folate, Vitamin E, Serum Copper, Ceruloplasmin and 24 hours-urinary Copper were normal. Vasculitic work up (ESR, CRP, ANA, Anti-dsDNA, ENA, RF, ANCA, and Complement) and paraneoplastic work up (Anti-Hu, Anti-Ri, AFP, PSA, CEA, CA 125, CA 153 and CA 19-9) were normal. Hepatitis C, B and HIV screening was negative. Lumbar puncture showed normal CSF protein and glucose with no cells or evidence of infection.
Upper GI endoscopy was repeated and biopsy ruled out any malignancy.
The clinical presentation of our patient showed a combination of pyramidal signs and loss of deep sensation especially in the lower limbs. Cervical myelopathy involving the dorsal column was evident on MRI. Laboratory studies showed macrocytic anemia with low cobolamin levels associated with high Homocysteine and MMA levels, which were diagnostic of Subacute Combined Degeneration of the Cord (SCD) secondary to vitamin B12 deficiency.
Investigations were done to determine the cause; Anti-parietal cells Abs and Anti-IF Abs were negative, however, serum Gastrin was highly elevated: 499ng/L (13-115). He was diagnosed with Pernicious Anemia (PA) based on the presence of macrocytic anemia, vitamin B12 deficiency and elevated serum Gastrin level despite negative Anti-IF Abs (IFA-Negative PA).
The patient was given intramuscular injections of vitamin B12 injection daily (1000mcg/day) for 2 weeks, followed by weekly intramuscular injections (1000mcg/week).
His blood cell count and blood film returned back to normal within 2 weeks of treatment initiation. Serum B12, homocysteine and MMA returned back to normal within 3 months. His motor strength, gait and balance showed subsequent marked steady improvement at 1-month, 3-months and 6-months follow- up visits. MRI of the cervical spine with contrast was repeated after 3 months of treatment and showed complete resolution of the cervical lesion (Figure 5).
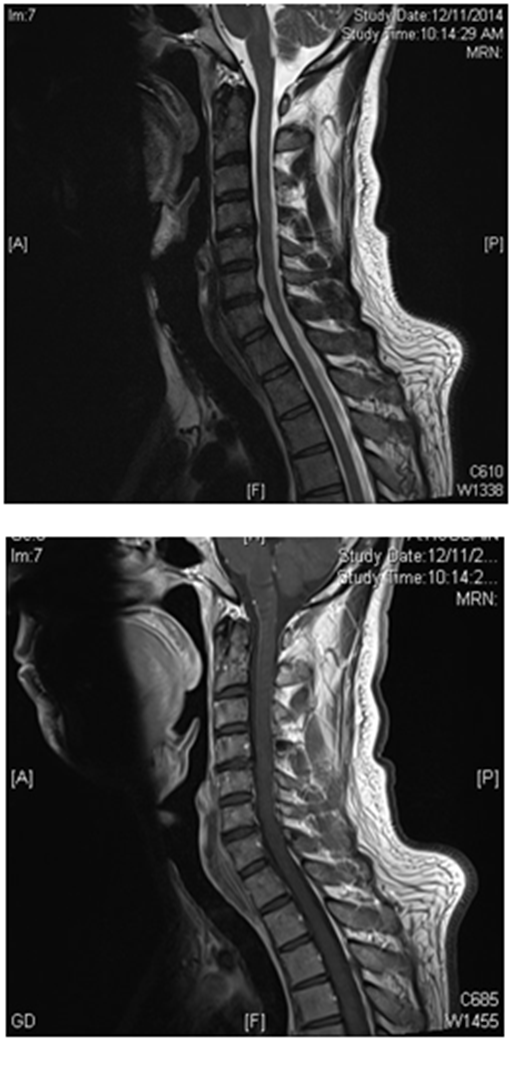
Figure 5 Follow up MRI Cervical spine (sagittal view) with and without contrast showing complete resolution of the cervical myelopathy.
Hyperpigmentation of the skin and the hair condition showed marked improvement within 6 months of treatment along with increase in his BMI to 24 (Figure 6).
His memory showed trivial improvement with persistence of lack of concentration and forgetfulness. MMSE after 6 months of treatment was 28.
Vitamin B12 is a water-soluble essential vitamin. It is also called "cobalamin" because it contains the metal cobalt. Vitamin B12, along with folate are essential for the synthesis of DNA during cell division thus it is important in formation of red blood cells and maintenance of a healthy nervous system as well as the skin.1
Vitamin B12 is not synthesized in the body and is found mainly in meat, eggs, and dairy products, as it is synthesized by microorganisms such as bacteria. It also lacks a reliable plant source. Daily requirements are about 1-3μg while body storage is around 2-5 mg which are sufficient for 3-4 years if supplies are completely cut off.2
Vitamin B12 is essential for three enzymatic processes: the conversion of homocysteine to methionine; the conversion of methylmalonic acid to Succinyl coenzyme A; and the conversion of 5-methyltetrahydrofolate to tetrahydrofolate (THF), a process necessary for DNA synthesis and red blood cell production. Thus its deficiency will lead to elevation in both homocysteine and methylmalonic acid.1,2
Vitamin B12 deficiency occurs either due to poor intake or decreased absorption. Poor intake is a rare cause although it can occasionally be seen in strict veganism. It typically results from Pernicious Anemia (PA), malabsorption (from sprue, enteritis, or infection with Diphyllobothrium latum), atrophic gastritis, surgical resection of the terminal ileum (often with Crohn's disease), Celiac disease, following Bariatric surgeries and can also be seen in infants from a mother with vitamin B12 deficiency. A very common cause of cobalamin deficiency is the widespread use of drugs as Metformin, proton-pump inhibitors and gastric acid-blocking agents, especially in the aging population.3
Vitamin B12 deficiency can be classified into two states: Clinical and Subclinical. Clinical deficiency manifests with hematologic or neurologic signs and symptoms, cobalamin levels less than 200 picograms per milliliter (pg/mL), and elevated levels for Homocysteine and Methylmalonic acid (MMA). Subclinical deficiency includes absent signs and symptoms, although some patients might have subtle changes on neurologic examination; low to low-normal Cobalamin levels (200–350 pg/mL); and at least one metabolic abnormality: elevated homocysteine or elevated methylmalonic acid.4,5
The first abnormality is usually sensory impairment, most often presenting as distal and symmetrical parasthesia of the lower limbs and frequently spreading to the hands, and accompanied by varying degrees of ataxia.6
The most common neurological signs on examination are diminished vibration sense and proprioception in the legs and present in almost all patients with associated positive Romberg sign. Loss of superficial sensation is usually present in a glove and stocking pattern. Sensory level is not usually present. Deep tendon reflexes (DTRs) may be exaggerated, diminished, or absent depending on the relative involvement of the cord.
Corticospinal tract involvement is common in more advanced cases; motor involvement and spastic paraparesis may occur, accompanied by autonomic bladder, bowel, or sexual symptoms. Plantar response is usually up-going in SCD.7
MRI is an important and sensitive tool in diagnosis of SCD; on sagittal images, a vertical segment can be seen at the posterior aspect of the spinal cord and B12 deficiency should be suspected in cases with longitudinally extensive transverse myelitis (LETM). On axial images, bilateral paired areas of T2 hyperintensity are seen as an “inverted V” or “inverted rabbit ears” (Figure 4) in the dorsal columns. Lateral column involvement is seen in severe cases. Contrast enhancement is uncommon. Signal abnormalities are usually reversed after treatment within a few months. MRI of the brain may show abnormal signal intensity on T2-weighted images in the cerebral white matter.8
Electrophysiological studies (NCS & EMG) usually show evidence of axonal peripheral neuropathy. Evoked potentials abnormalities may be the first electrodiagnostic finding, even in asymptomatic patients with normal neurologic examination; abnormal SSEP denotes myelopathy while abnormal VEP denotes optic neuropathy.9
Psychiatric manifestations are common. They include major depressive disorder, personality changes and psychosis. Memory changes are prominent feature and can lead to development of mild cognitive impairment (MCI) or Dementia.10
The independent association between vitamin B12 deficiency and dementia is well documented though out the medical literature.11 Cognitive decline is attributed to the duration of deficiency and to the levels of homocysteine and MMA; the longer the duration and the higher the levels, the more severe the symptoms will be.11,12
There is a controversy in literature regarding improvement of the cognitive decline with vitamin B12 supplementation as several intervention studies failed to find a statistically significant improvement in cognitive function.13
In one review including data from four vitamin trials, there was no evidence that folate and/or B12 had significant effects on cognitive function.14,15 However, there are several major limitations for all available studies including small number of subjects, short duration of treatment and long duration of illness.
Hematological manifestations are mainly related to anemia and include pallor, fatigue, palpitations and tachycardia. Hematologic abnormalities may be absent at the time of neurologic presentation. Interestingly, hematologic and neurologic manifestations are occasionally dissociated; an inverse correlation in the severity of both manifestations has been suggested. In patients with neuropsychiatric abnormalities, 28% lack anemia or macrocytosis.16
Vitamin B-12 deficiency produces the classic picture of macrocytic anemia, with a mean corpuscular value (MCV) greater than 100 fL. The MCV usually correlates with estimated vitamin B-12 level; normal MCV of (80-100 fL) indicates less than 25% probability of vitamin B-12 deficiency, MCV of (115-129 fL) indicates a 50% probability and MCV greater than 130 fL indicates a 100% probability. However, up to 28% of affected patients may have a normal hemoglobin level, and up to 17% may have a normal mean corpuscular volume.17
Blood study typically shows macro-ovalocytosis, anisocytosis, and poikilocytosis, as well as basophilic stippling of the erythrocytes. Reticulocytic count can be low or normal. Hypersegmentation (>5% of neutrophils with >5 lobes or 1% with >6 lobes) of polymorphonuclear cells may occur without anemia. Leucopenia and thrombocytopenia are observed in approximately 50% of patients and they are correlated with the severity of the anemia.
Serum indirect bilirubin and lactate dehydrogenase (LDH) may be elevated because PA can have a hemolytic component.
Hematological parameters are the first to improve with B12 supplementation; LDH falls within 2 days, reticulocytosis starts in 3-4 days and peaks at 1 week, hypersegmented neutrophils disappear in 1-2 weeks while hemoglobin concentration rises in 10 days and returns to the reference range in 8 weeks.3
Dermatological manifestations of Vitamin B12 deficiency include hyperpigmentation especially in dark-skinned patients, lemon-yellow waxy pallor, brittle hair with premature whitening, glossitis, angular stomatitis and associated vitiligo. These cutaneous changes are due to increased melanin synthesis in the basal layer of the epidermis as cobalamin decreases the level of reduced-type glutathione, which normally inhibits the rate-limiting enzyme of melanin synthesis tyrosinase. Hyperpigmentation may be the first manifestation of vitamin B12 deficiency. The pattern of hyperpigmentation is generalized with accentuation in flexural areas, palms and soles. It may also be accentuated in areas of pressure, such as the terminal phalanges, knees, and elbows. Also areas of blotchy darkening and linear streaks may often be seen.
Our patient's manifestations were secondary to undiagnosed pernicious anemia which is a chronic, autoimmune disease characterized by vitamin B12 deficiency secondary to failure of gastric parietal cells to produce intrinsic factor (IF) in the stomach; it accounts for 75% of cases of vitamin B12 deficiency. Patients could be either IFA-positive (70%) or IFA negative (30%), the latter of which true in our case. They may block the formation of the cobalamin-IF complex or block its binding with cublin. Other antibodies are directed against parietal cell hydrogen-potassium adenosine triphosphatase (ATPase). Chronic atrophic gastritis and gastric ulcers are a common complication and there is an increased likelihood for such patients to develop cancer stomach.18
PA should be ruled out in any patient with low vitamin B12 levels by measuring antibodies against IF first. IF antibodies are present in 70% of patients. They are more specific but less sensitive than Anti-parietal cell antibodies which are present in 90% of cases. If either antibody is positive, then the diagnosis of PA is confirmed and further testing is not required, but if both antibodies are negative, then serum Gastrin level should be assessed for achlorhydria, which is associated with PA in 90% of cases. If these are elevated, then the diagnosis is most likely PA. If all such results are normal, then the Schilling test should be performed.19,20
In our patient both antibodies were negative, however, serum gastrin was highly elevated which is highly sensitive in diagnosis of PA.
Findings of decreased serum cobalamin, elevated methylmalonic acid (MMA) and\ or elevated homocysteine can be used to make a diagnosis of vitamin B12 deficiency. B12 levels are not a reliable indicator and are usually normal. One study of 406 patients with known vitamin B12 deficiency showed that 98.4 % of patients had elevated serum methylmalonic acid levels and 95.9 % had elevated serum homocysteine levels (defined as three standard deviations above the mean). When both methylmalonic acid and homocysteine levels are used for diagnosis, there is a sensitivity of 99.8 %. If both metabolites are normal, vitamin B-12 deficiency is effectively ruled out.17
Several treatment regimens have been proposed, most frequently being Cobalamin 1000 mcg IM/SC daily for 5 days followed by 1000 mcg/wk for 5 weeks, then 100-1000 mcg/month for life. Oral administration of high-dose vitamin B12 (1 to 2 mg daily) was found to be as effective as intramuscular administration in correcting the deficiency, regardless of etiology because 1% of cobalamin is absorbed by passive diffusion, so 1000 mcg tablet daily will lead to absorption of 10 mcg, which exceeds the 2-mcg recommended daily allowance (RDA) requirement.7
Clinical improvement with treatment is observed mostly in the first 2 months but may take up to 6 months. Hematological profile improves first within 2 weeks followed by neurological and dermatological symptoms. Cognitive impairment is poorly responsive to treatment in the short-term. Factors favoring a good prognosis are: younger patients with less severe disease and short duration of illness. Clinically, absence of sensory level, absent Rhomberg sign, and flexor planter reflex were associated with good prognosis.7,21 Radiologically, involvement of less than 7 spinal segments and enhancement, but not cord atrophy, was associated with better prognosis.22
Vitamin B12 deficiency is a common but under-diagnosed clinical condition that needs to be highlighted. It can present with neurological, psychiatric, hematological and/or dermatological manifestations. It is not common to see the whole systems affected together early in the course of the disease. Early diagnosis and treatment may reverse potentially permanent symptoms and signs and can prevent the disability that would otherwise ensue if left untreated. Pernicious anemia is one of the most common causes of vitamin B12 deficiency and should be ruled out in all cases even if autoantibodies against intrinsic factor and parietal cells are negative.
None.
None.

©2015 Al-Hashel, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.