Journal of
eISSN: 2373-6410


Case Report Volume 13 Issue 6
Department of Neurosurgery, Hassan II university hospital of Fez, University Sidi Mohammed Ben Abdellah, Fez, Morocco
Correspondence: Marouane Hammoud, Assistant professor, department of neurosurgery, Hassan II university hospital of Fez, university Sidi Mohammed Ben Abdellah, Fez, Morocco, Tel 0661491896
Received: November 23, 2023 | Published: December 28, 2023
Citation: Hammoud M, Alaoui NK, Benzagmout M, et al. Primary hydatid cyst of cerebellar vermis: a pediatric case report with updated surgical management. J Neurol Stroke. 2023;13(6):176‒178. DOI: 10.15406/jnsk.2023.13.00569
Introduction: Hydatid cyst is a rare parasitic disease caused by Echinococcus granulosus larvae, with the liver being the most commonly affected organ, followed by the lungs and spleen. Brain involvement, is uncommon, representing approximately 1-2% of reported hydatid cyst cases worldwide. Mostly located in the supratentorial region. Cerebellar location is even rarer.
Case description: A 3-year-old boy was referred to our department exhibiting signs of increased intracranial pressure, ataxia, and diplopia. Brain computed tomography (CT) scan and magnetic resonance imaging (MRI) revealed a well-defined intra-axial unilocular cystic lesion in the cerebellar vermis without contrast enhancement, indicative of a hydatid cyst. Surgical management, utilizing Dowling’s technique, resulted in a very favorable outcome with the patient achieving full recovery.
Conclusion: This case underscores the importance of meticulous surgical treatment to prevent cyst rupture, as this complication can significantly worsen the overall outcome.
Hydatidosis, a zoonotic disease caused by the adult and larval stages of the parasite Echinococcus granulosus, stems from contact with animals hosting the parasite, primarily canines such as dogs, wolves, and foxes, which serve as definitive hosts. Humans inadvertently become intermediate hosts through the consumption of contaminated food or direct contact with dogs.1,2
Echinococcus eggs traverse the human intestinal mucosa, entering the portal system and disseminating hematogenously to various organs. The liver is the most commonly affected organ, accounting for 75%, followed by the lungs at 15%.
Involvement of the central nervous system is rare, representing 1–2% of all Hydatid disease cases, with the majority occurring in children and young adults, boasting prevalence rates of approximately 50–70%.3 Hydatid disease remains a significant public health concern in areas with a high prevalence of sheep and cattle raising, such as Australia, East Africa, South America, Eastern Europe, and the Middle East, with prevalence rates ranging from 1.2 to 23 per 100,000 inhabitants.4
This case report highlights an unusual occurrence—a primary hydatid cyst located in the cerebellar vermis of a 3-year-old boy, confirmed through histopathological examination.
General
A 3-year-old boy with an unremarkable medical history presented to our hospital with a four-month history of intermittent headaches, escalating over the previous week. Vomiting, neck stiffness, and diplopia accompanied the headaches. Neurological examination revealed cerebellar ataxia, without any focal neurological abnormality, cranial nerve deficit, or papilledema. His head circumference was within the normal range. Routine blood analyses yielded normal results.
Imaging and diagnosis
Brain computed tomography (CT) scan and magnetic resonance imaging revealed a well-defined rounded cystic lesion in the vermis, exerting mass effect on the brainstem and fourth ventricle. The lesion appeared hypodense on CT scan, hypo-intense on T1-weighted images, hyper-intense on T2 with slight surrounding edema, better visualized on fluid-attenuated inversion recovery sequences. No enhancement of the cyst wall was observed (Figure 1).
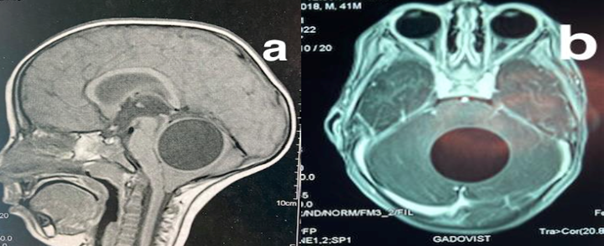
Figure 1 (A) Midsagittal T1-weighted MRI showing a vermian rounded cystic lesion. (B) Axial T1-weighted MR image with godalinium showing no contrast enhancement.
Diagnostic considerations leaned towards a pilocytic astrocytoma and cerebral hydatid cyst. Given the epidemiological context (contact with dogs and rural living), cerebral hydatid cyst emerged as the more plausible diagnosis. Additional investigations, including chest X-ray and abdominal ultrasonography, revealed no abnormalities. Serological tests for hydatid disease returned positive.
Treatment
The patient underwent total cyst excision via a suboccipital approach. Following dural exposure, a vermian incision was made to access the cyst wall. A distended, dense cystic membrane was observed. Using a flexible probe, we facilitated delivery by hydro pulsion, following the Dowling–Orlando technique. The cyst was completely removed with an intact capsule by irrigating saline between the cyst wall and brain interface (Figure 2). Duroplasty was performed.

Figure 2 (A) Per operative image showing the cyst wall after the vermian incision (B) Intact cyst removal by irrigating saline between cyst wall and brain interface.
Postoperative
The postoperative period was uneventful, and the patient's symptoms markedly improved within one week. Histopathologic examination confirmed the diagnosis of a hydatid cyst. Albendazole was initiated at a dosage of 10mg/kg/day and continued for three months. The patient was discharged on the fifth postoperative day. In the most recent examination conducted 1 year post-surgery, the patient remains stable. Brain CT scan showed no reccurence (Figure 3).
Cerebral hydatid cyst, a rare occurrence primarily impacting children and young adults1,5 is predominantly found supratentorially in the distribution of the middle cerebral artery. Infratentorial locations are uncommon, comprising only 1% of cerebral cases.3,6 Distinguishing between primary and secondary cysts, the former arises from direct larvae infestation in the brain, while the latter emanates from other primary sites like the liver or lungs.7
Clinical manifestations vary based on location, size, and the duration of symptoms, with the latter serving as an independent predictor of the outcome.3 Characterized by slow growth, approximately 1 cm/year, this benign lesion presents distinct features on brain CT scan and MRI, appearing as a large, rounded, unilocular, thin-walled cyst with hypo-signal contents on T1-weighted images and hyper-signal on T2-weighted images. The absence of contrast enhancement and edema is typical, and calcification on CT scan may indicate parasite death, while irregularities in the cyst wall may suggest prior rupture.9–11
Differential diagnoses encompass arachnoid cysts, porencephalic cysts, and cystic tumors like pilocytic astrocytoma.12 While serological tests provide valuable diagnostic and postoperative follow-up insights, their accuracy, ranging between 60-90%, necessitates complementary assessments.13
Surgical removal remains the cornerstone for treating intracranial hydatid cysts, with the goal of intact cyst excision guiding the choice of technique.14,15 The Dowling–Orlando technique proves valuable for this purpose, involving a large craniotomy, microsurgical dissection, an inclined head position, and saline irrigation.Intraoperative tools such as ultrasound or neuronavigation assist in guidance, especially for deep-seated cysts.16
Aspiration followed by cyst wall removal is an other effective technique but carries a risk of rupture and dissemination, limiting its use only for the treatment of deep-seated hydatid cysts such brainstem or cerebellopontine cistern when intact removal is not possiblie.17 While surgical extirpation remains the primary modality, the role of antihelminthic therapy is indispensable in specific scenarios, including recurrence, hydatid dissemination, inoperable lesions, or intraoperative cyst rupture.18,19
Albendazole, administered at a dosage of 12-15 mg/kg/day for three months, has demonstrated efficacy in reducing recurrence rates post intact surgical removal. Nevertheless, vigilant monitoring for hepatotoxicity, through regular transaminase tests during the initial three months, is imperative.20,21
Hydatid cyst should be considered in the differential diagnosis for cystic lesions of the posterior fossa. A heightened clinical suspicion and consideration of this parasitic disease are pivotal for ensuring effective surgical management, given the potential for dramatic consequences if not well managed.
None.
The authors declare no conflicts of interest.

©2023 Hammoud, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.