Journal of
eISSN: 2373-6410


Review Article Volume 12 Issue 4
Department of Neurology, University Hospital, Belgium
Correspondence: De Reuck J, Ryvissche park 16, 9052 Zwijnaarde. Belgium, Tel 0032 (0) 474 652076
Received: June 14, 2022 | Published: July 5, 2022
Citation: Reuck JD. The Wernicke-Korsakoff encephalopathy: An updated review. J Neurol Stroke. 2022;12(4):79-82 DOI: 10.15406/jnsk.2022.12.00505
Wernicke's encephalopathy is responsible for an acute neuropsychiatric syndrome that is associated with significant morbidity and mortality. It is most frequently due to alcoholism but can also be the result of chronic diseases, mainly systemic tumours, leading to thiamine deficiency. In the non-thiamine treated patients Korsakoff’s syndrome is the residual complication of the encephalopathy. The clinical diagnosis of Wernicke encephalopathy in alcoholics requires two of the following four signs: dietary deficiencies, eye signs, cerebellar dysfunction, and either disturbed mental state or mild memory impairment. These symptoms are less specific in the non-alcoholic patients. Korsakoff's syndrome is the residual condition in none thiamine treated patients. It is predominantly characterized by global amnesia, and in the more severe cases also by cognitive and behavioral dysfunction. Magnetic resonance imaging of the brain can detect the specific lesions and be helpful for the diagnosis. Treatment with 2500 mg thiamine intravenously is recommended as soon as possible.
Keywords: Wernicke encephalopathy, Korsakoff’s syndrome, thiamine treatment, alcohol abuse, dietary deficiencies
The first most complete review on the Wernicke-Korsakoff encephalopathy was published by Victor, Adams and Collins in 1971. They described not only the clinical features but also the neuropathological lesions in more than 500 post-mortem brains. The incidence of Wernicke’s encephalopathy (WE) and Korsakoff (KS) could at that time not be stated with precision. However in a series of 1459 autopsies in adults 2.7% were found to have lesions characteristic for WE and/or KS.1
Wernicke's encephalopathy (WE) was found associated with significant morbidity and mortality.2 WE and Korsakoff syndrome (KS) were termed together as Wernicke-Korsakoff syndrome (WKS) because they were found distinct but yet overlapping neuropsychiatric disorders associated with thiamine deficiency.3
First described in 1881 by Carl Wernicke, WE continued to be an unrecognized and often a misunderstood disease. He described the nystagmus, the abducens and the conjugate gaze palsies, the ataxia of gait, and the mental confusion. He called this disease polioencephalitis hemorrhagica superioris. The symptom complex of KS was described in 1887 by the Russian neurologist and psychiatrist Sergei S. Korsakoff. He defined it as an amnestic or amnestic-confabulatory psychosis and a psychosis polyneuropathia.4 In a necropsy study in Western Australia, the incidence of WE was 2.8%.5
Apart from chronic alcoholism as most common cause of WE, a lot of other conditions causing malnutrition and decreasing thiamine absorption were considered as predisposing factors.6 The first cases of non-alcoholic WE have been presented in 1980 at the first European Neuropathology meeting in Vienna.7 Younger age seemed to protect against the mental alterations and higher body mass index against the eye movement disorders.8
Abnormalities in the thiamine dependent enzyme transketolase provide evidence of a high incidence of thiamine deficiency as well as of disturbed thiamine metabolism in chronic alcoholics, which are likely to be caused by the reduced vitamin B1 intake as well as an impairment of its absorption. Glutamate neurotoxicity is also a major cause of the brain lesions in WE. Glutamic acid decarboxylase, an enzyme mainly confined to the central nervous system, protects most regions of the brain from glutamate that accumulates when the activity of alpha-ketoglutarate dehydrogenase, a thiamine-dependent enzyme complex, is reduced.9 Specifically, patients who show rapid weight loss are vulnerable for the development of WE. Eating disorders can also lead to WE.10
Not eating a balanced diet or fasting can cause neurological complications after severe vitamin B1 malnourishment, although the precise signs and symptoms of WE are not always clear.11 Also, the extensive use of diuretics can lead to WE by increasing the urinary thiamine excretion depending on the urinary flow rate.12
A pilot study of 31 consecutive alcohol related deaths over an 8-month period reveals the presence of histologically diagnosed WE in 17 cases. Analysis of the clinical records of these patients show that a disturbance of the mental state is the commonest finding and that the other neurological signs such as ataxia, eye movement disturbances and peripheral neuropathy are present in only 2 of the 17 cases.13
The pathological findings include recent petechial and local hemorrhages in the mamillary bodies, and the periventricular regions around the third and fourth ventricles and the aqueduct (Figure 1). Under light microscopy the proliferation and the dilatation of the capillaries are particularly prominent in the mamillary bodies and pericapillary hemorrhages are present in the periventricular regions (Figure 2). Neuronal losses are found only in the medial dorsal nucleus of the thalamus and in the inferior olive. Luxol blue staining demonstrates demyelination and gliosis in those areas.14
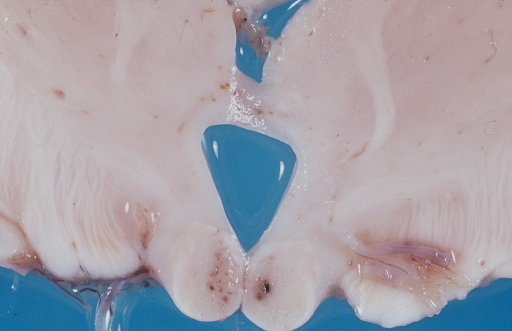
Figure 1 Macroscopic post-mortem coronal section of the cerebral hemispheres with Wernicke’s encephalopathy in a 32-year old woman, who has been treated for centroblastic-centrocytic lymphomas.
Note the small bleeds in both mamillary bodies.
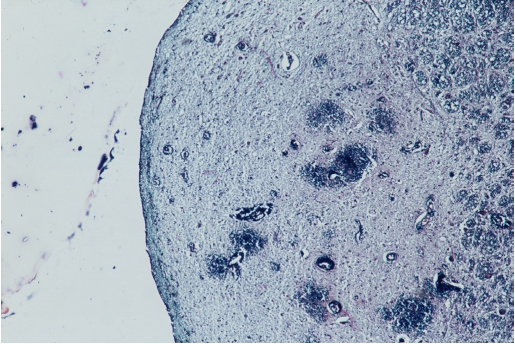
Figure 2 Histological luxol fast blue staining of a mamillary body in the same 32-year old woman.
Note the abundant perivasculair bleeds around many capillaries.
In recent years, however, astrocytes are considered as a main target in the pathophysiology of thiamine deficiency. Glutamate uptake, and levels of the astrocytic glutamate transporters EAAT1 and EAAT2 in thiamine deficiency and WE, are to be considered as an excitotoxic event, along with the GABA transporter subtype GAT-3, and changes in other astrocytic proteins including glial fibrillary acid one and the glutamine synthetase. Lactic acidosis, changes in the water channel protein anti-aquaporin 4 and brain edema are also a focus of attention in relation to astrocyte dysfunction, while involvement of oxidative stress and inflammatory processes, along with white matter injury in terms of excitotoxicity are other key issues to be considered.15
WE should be suspected in all clinical conditions that could lead to thiamine deficiency. The clinical diagnosis of WE should take into account the different presentations of clinical signs between alcoholics and non-alcoholics, although the prevalence is higher in alcoholics. Apart from alcoholism the most common causes of WE are a lot of other conditions causing malnutrition and decreasing thiamine absorption. The clinical diagnosis of WE in alcoholics requires two of the following four signs; dietary deficiency, eye signs, cerebellar dysfunction, and either an altered mental state or a mild memory impairment.16
A necropsy study shows that 80% of patients with the WKS are not diagnosed as such during life. Review of the clinical signs of these cases reveals that only 16% present the classical clinical triad (mental status changes, ocular symptoms, and ataxia) and 19% has no specific clinical signs.17
Non-alcoholic WKS can occur during cancer treatment and manifests clinically mainly as a delirium. The patients present cognitive dysfunction, most commonly impaired alertness, attention, and short-term memory.18 In adults the most frequent tumors are those of the lymphoid-hematopoietic systems. Most of these patients are not underweight and have normal serum concentration of vitamin B12 and folate. A variety of mechanisms might predispose to thiamine deficiency and WKS in cancer.19
Other rare causes of WKS are Crohn’s disease and ulcerative colitis. In 12 cases, partial or complete bowel resection is one of the etiologies of thiamine depletion. In nine other cases, thiamine was not supplemented intramuscularly or intravenously while parenteral nutrition or glucose was given to the patients.20Also, a WE case due to an acute pancreatitis is described. It appears during the restoration stage of the pancreatitis.21 WE is a rare complication of hyperemesis gravidarum. Subsequent treatment with thiamine led to rapid improvement.22
A 57-year-old male patient with gastric carcinoma who underwent a radical distal gastrectomy type II + Braun and who developed refractory arterial hypotension is found at necropsy to have WE.23 WE is also observed in two non-alcoholic homosexual patients with acquired immunodeficiency syndrome patients.24 In children the most frequent underlying disorder is malignancy. In more than half of them they present with the WE clinical triad of mental status changes, ocular signs, and ataxia at neurologic onset. Some patients died undiagnosed. 16 recovered with thiamine therapy, However half of them had remaining neuropsychiatric sequels and two died of infection soon after thiamine replacement was initiated.25 Poor nutrition in children is the most frequent under diagnosed cause of WE and is only proven by the post-mortem neuropathological examination.26
Neuroradiological examination with magnetic resonance imaging (MRI) is a valuable tool for the diagnosis of acute WE and enables the in vivo tracking of the progression of the brain pathology from the acute pathological phase to resolution with thiamine treatment or to progression to KS without treatment.27
In the acute WE, MRI shows a high signal intensity on T2-weighted images in periaqueduct and medial thalamic regions. In a few patients with alcoholism, vermian and mammillary body atrophies and third ventricular enlargements are noted. In the chronic phase of WE, the T2 hyper intensity disappears but mammillary bodies and cerebellar vermis become atrophic and third ventricular enlargements become evident. The contrast enhancement can sometimes be very intense in the early WE cases.28 The high signal intensity on T2-weighted images disappears as early as within 2 days, and atrophic changes appear as early as 1 week.29
Contrast enhancement in the mammillary bodies and thalamus is a typical finding in acute alcohol-related WE. Atypical MR imaging findings, on the other hand, characterize the non-alcoholic patients. Alcohol induced WE has on MRI more contrast enhancement in the mamillary bodies and thalami than in non-alcoholic WE. This supports the hypothesis that alcohol may contribute to increased blood-brain barrier permeability.30
Diffusion-weighted imaging abnormalities of the splenium of the corpus callosum can be demonstrated in some cases of WE.31 In a patient with non-alcoholic starvation induced atypical WE symmetrical lesions in substantia nigra are observed, in addition to the classical neuroradiological findings.32 Another non-alcoholic atypical case of WE presents with reversible symmetrical lesions in the cranial nerves nuclei and in the cerebellum as well as the classic MRI findings.33
Symmetric basal ganglia alterations with involvement of the putamen are only observed on MRI in children.34
Even in a patient with alcohol-induced WE without visual lesions on cranial MRI, brain single photon emission computed tomography can reveal bilateral frontal and fronto-parietal hypoperfusion as well as right basal ganglia hypoperfusion.35
In a series of positron emission tomography scans of WE patients a significantly reduced flumazine ligand influx and distribution volume is observed. Low cerebral metabolic rates for glucose are mainly observed bilaterally in the medial frontal lobes, including the superior frontal gyri and the rostral cingulate gyri and also in the superior cerebellar vermis.36
A retrospective analysis of initial fluorodeoxyglucose-positron emission tomography images using a voxel-wise statistical method also reveals glucose hypo-metabolism in the diencephalon and basal forebrain. Follow-up for 5 years after the initial evaluation shows recovery of glucose metabolism in the two brain regions linked clinically to an improved cognitive function.37
WE is an acute neuropsychiatric syndrome associated with significant morbidity and mortality.2 KS is the residual syndrome in not-thiamine treated patients who had suffered from WE. It is predominantly characterized by global amnesia, and in more severe cases also by cognitive and behavioral dysfunction.38
Upon short-term sobriety cerebellar choline and fronto-mesial N-acetyl-aspartate become significantly augmented. Automatically detected global brain volume gain amounts up to nearly two per cent on average spatially. This is mainly significant around the superior vermis, the peri-mesencephalic and frontal brain lobes. It suggests that early brain recovery through abstinence does not simply reflect rehydration.39 Visual long-term memory, and verbal fluency improve slightly after two years, though they still remain within the pathological range.40
The literature indicates that thiamine should be prescribed at high dosages, as early as possible in high-risk alcoholic patients, by intravenous routes during 3 to 5 days and in a hospital setting.41 Based on the current literature high-dose thiamine (≥500 mg) appears to be safe and efficacious for the use in patients with suspected alcohol-related WE.42
Thiamine-related WE in cancer patients also improve following thiamine treatment, allowing patients to engage with their family and treatment teams prior to recovery or death.43 The risk of anaphylaxis after intravenous thiamine treatment is low, and lower than for many other drugs. The risk-benefit ratio for administration is favorable given the potential severity of brain damage in WKS.44
WKS is an under diagnosed and frequently a fatal disease. In a necropsy series in western Australia an incidence of WE was 2.8%. Only 20% of them have clinically been diagnosed as WE.45
There is no single, well-studied intervention proven to be effective as a primary treatment for the cognitive impairment in KS.46
The author has nothing to declare in relation to this article. No funding was received for the publication of this article.
None.
The authors declare no conflicts of interest.

©2022 Reuck. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.